What are proteoforms?
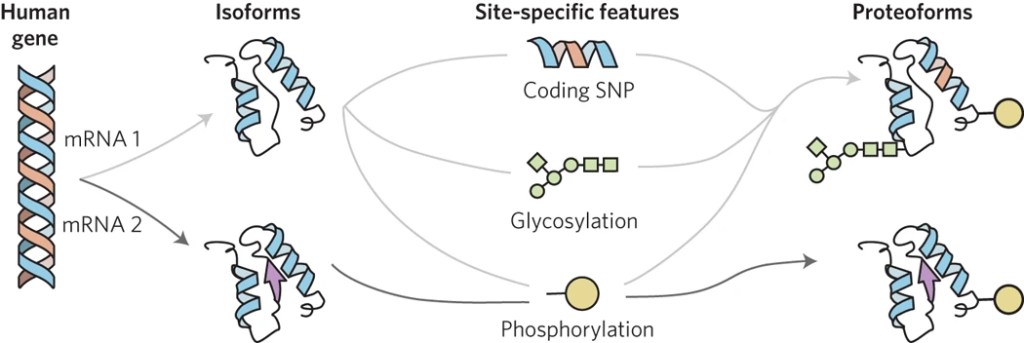
Proteoforms are the different forms of proteins produced from the genome with a variety of sequence variations, splice isoforms, and myriad post-translational modifications. They represent the functional units of proteins with precise primary structures and are more tightly connected to cellular functions, phenotypes, and diseases.
What is top-down proteomics?
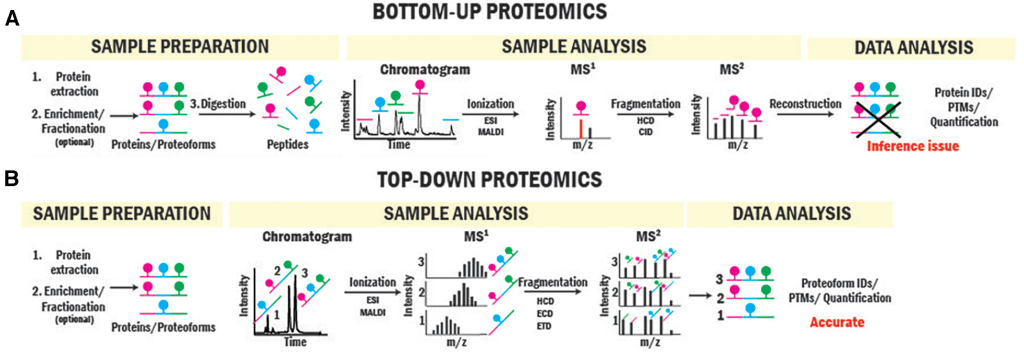
While peptide-based bottom-up proteomics identifies and quantifies proteins and post-translational modifications with great sensitivity and dynamic range, there is an inference challenge of mapping peptides to the proteoforms they originate from. Top-down proteomics addresses this issue by directly studying intact proteins using mass spectrometry. Proteoform sequencing can be achieved by further fragmenting intact protein ions within the mass spectrometers.
Connecting proteoforms to functions
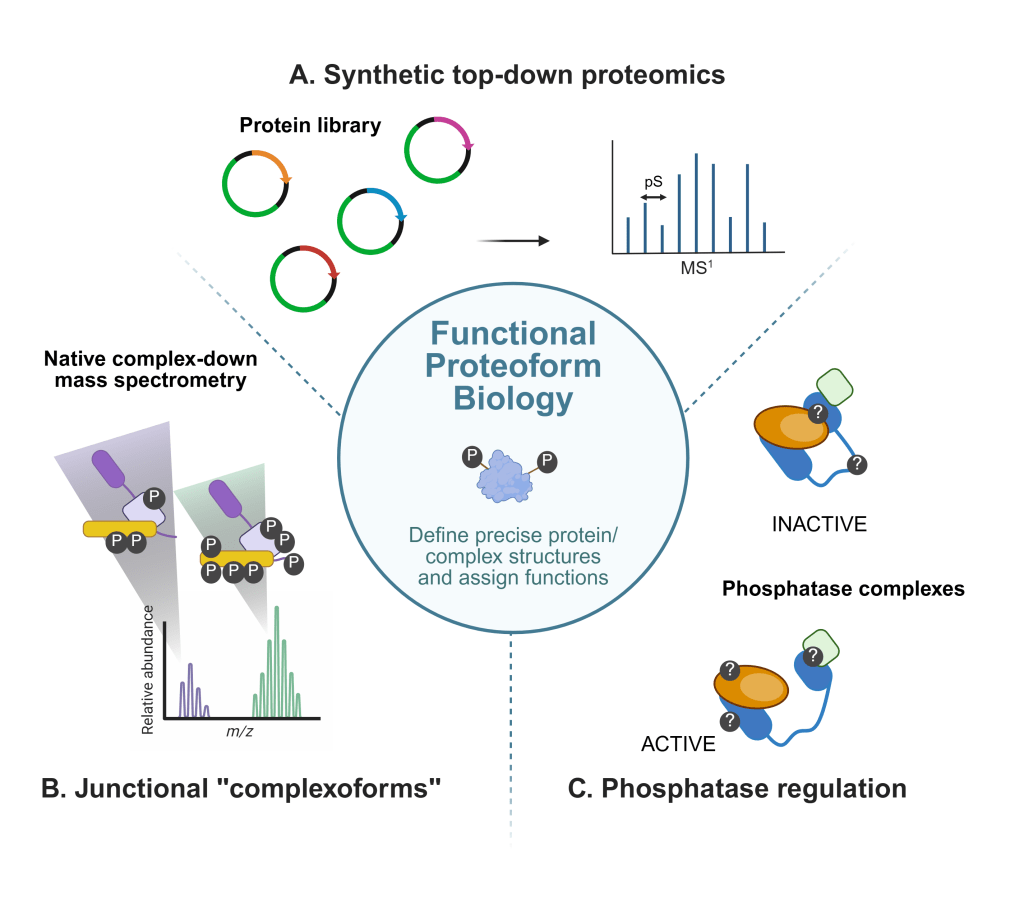
The Huang Research Group develops chemical and measurement strategies to understand how covalent modifications control protein structure and function in complex biological systems. We advance top-down proteomics, the mass spectrometric analysis of intact proteins and protein assemblies, to directly characterize proteoforms as chemically defined molecular species. By integrating chemical biology, quantitative mass spectrometry, and molecular engineering, we determine how phosphorylation and other post-translational modifications tune protein structure, stability, enzymatic activity, intermolecular recognition, and higher-order assembly.
Our research focuses on phospho-regulation in two interconnected systems: cadherin–catenin complexes that govern cell–cell adhesion and phosphatases that shape and maintain phosphorylation networks. We seek to establish quantitative protein structure–function relationships that explain how specific proteoforms encode distinct cellular states. By building tools to characterize intact protein chemistry in native contexts, our work connects molecular structure to signaling behavior and cellular mechanics, with particular relevance to epithelial barrier integrity, cancer-associated signaling, and other diseases driven by dysregulated phosphorylation.